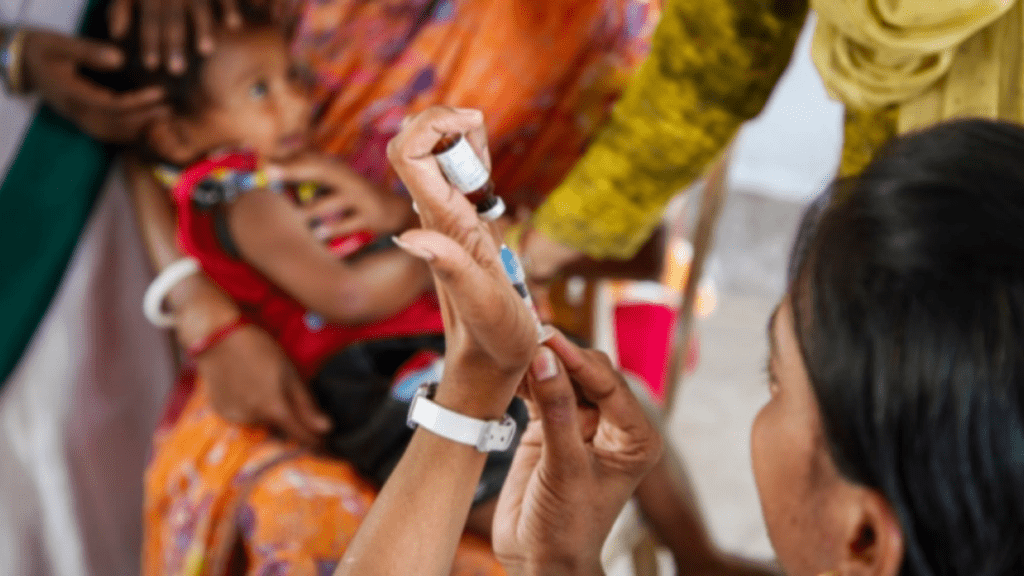
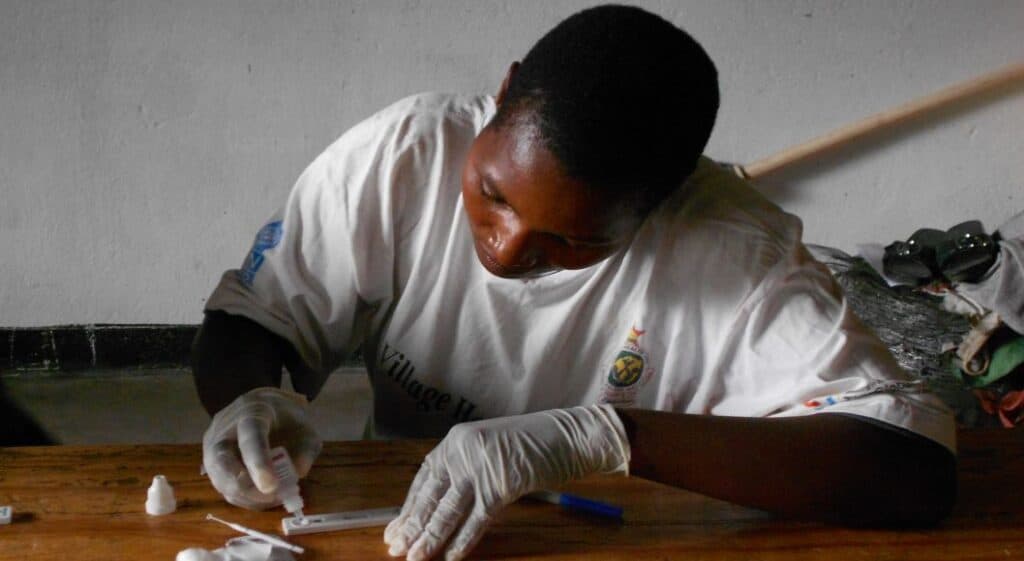
Globally, cervical cancer kills more than 250,000 women every year, 90 percent of whom live in low- and middle-income countries. When provided to young girls between 9 and 14 years old, the human papillomavirus (HPV) vaccine is the most effective way to prevent cervical cancer.
Since 2015, JSI has applied its immunization systems strengthening expertise to HPV vaccine demonstration projects, national introductions, implementation, and learning in 13 countries: Cameroon, DRC, Ethiopia, Kenya, Madagascar, Malawi, Mozambique, Niger, Nigeria, Sao Tome and Principe, Tanzania, Zambia, and Zimbabwe. Along the way, we have gained a deep understanding of HPV vaccine implementation and integration into national immunization programming, infusing country experience and learning into regional and global policy and technical guidance.
JSI’s HPV vaccine technical assistance (TA) approach is grounded in being country-led through trusted immunization experts working in close collaboration with the Ministry of Health’s Expanded Program on Immunization, Ministry of Education and/or other government programs, Gavi, the Vaccine Alliance (Gavi), WHO, UNICEF, and others. JSI helps countries strengthen programming to achieve high HPV vaccine coverage and sustainable programming. It supports evidence-based decision-making; Gavi HPV vaccine grant applications; HPV vaccine introduction and multi-age cohort planning and implementation; and post-introduction program strengthening and sustainability and learning. Within this, JSI specializes in national and sub-national microplanning; health worker training; social mobilization and communications; supply chain and logistics; national coordination, planning and advocacy; and supportive supervision and monitoring.