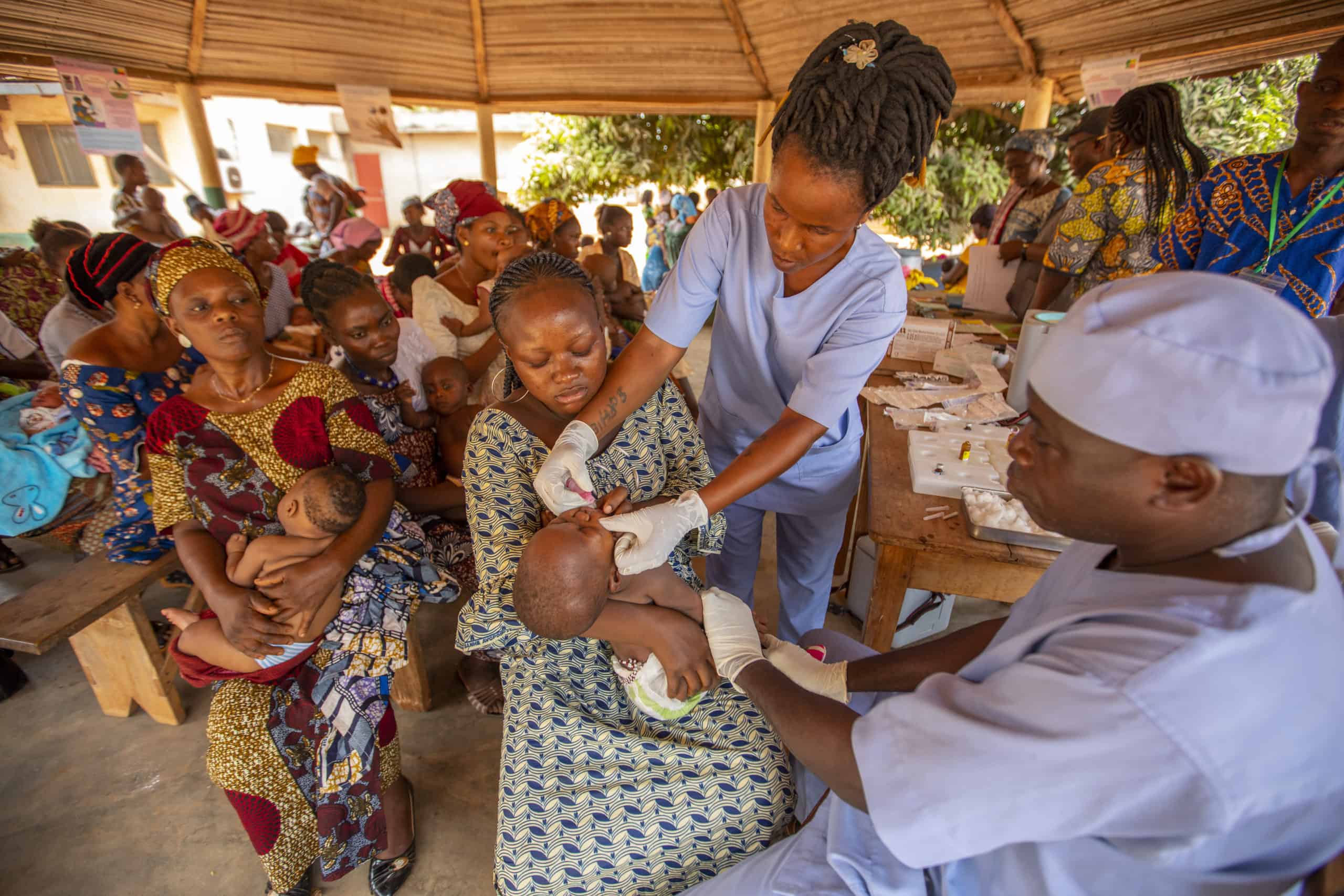
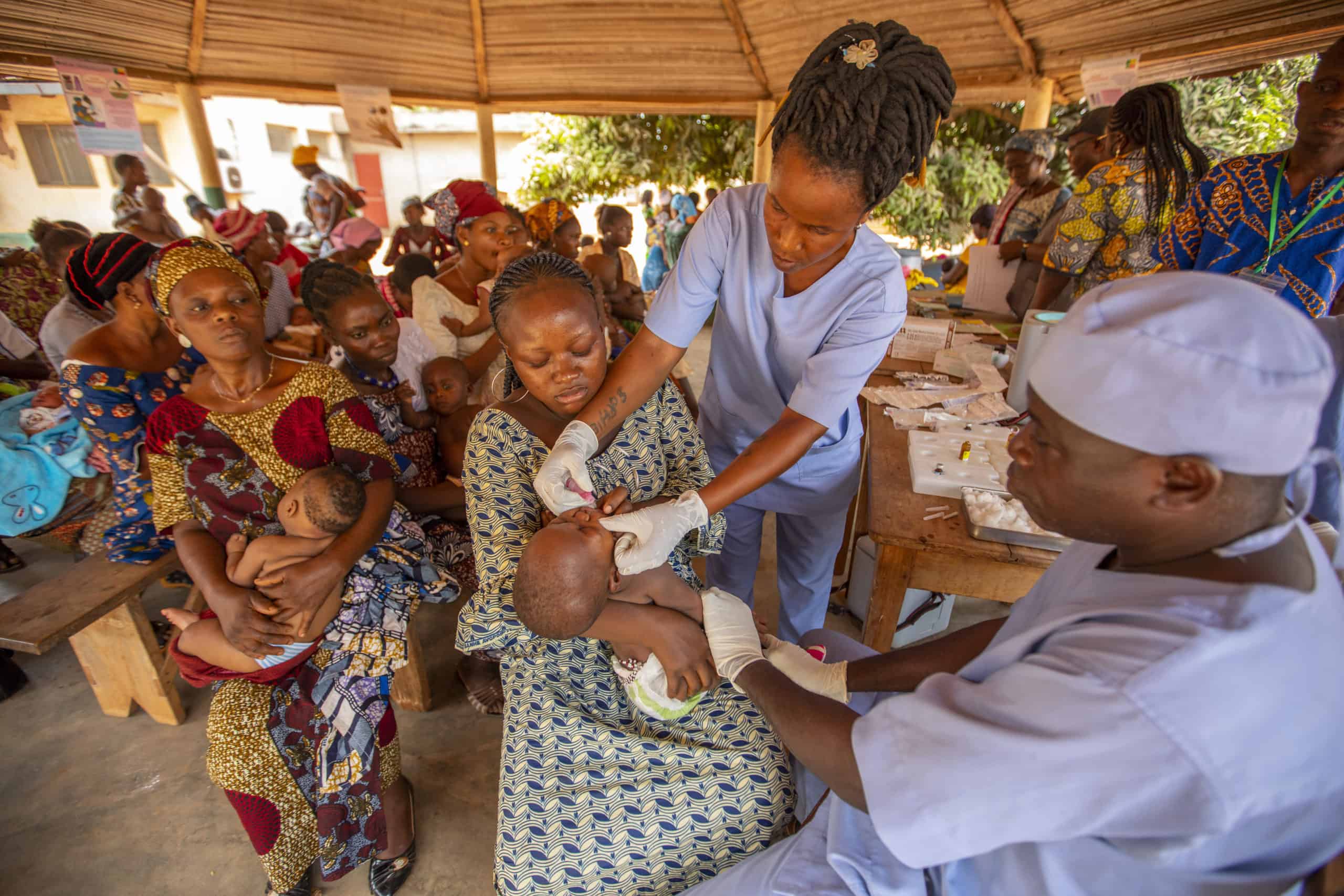
The Rotavirus Accelerated Vaccine Introduction Network (RAVIN) provided technical support to countries considering introducing the rotavirus vaccine into their national immunization programs. JSI worked closely with Ministries of Health and in-country partners in focus countries to support the successful introduction of the vaccine.
Diarrhea is one of the world’s leading killers of children, and rotavirus is the most common cause of severe diarrhea. Each year, rotavirus kills about 200,000 children and hospitalizes hundreds of thousands more, despite the availability of safe, effective rotavirus vaccines that can protect children from disease and death. Over 90 million infants worldwide lack access to the vaccine – tens of millions of whom live in Gavi-focus countries that have not yet introduced the vaccine into their health systems.
In 2015, the Bill & Melinda Gates Foundation awarded RAVIN to the Johns Hopkins Bloomberg School of Public Health’s International Vaccine Access Center (IVAC), in collaboration with JSI and the Centers for Disease Control and Prevention (CDC) to provide rotavirus vaccine support to focus countries: Afghanistan, Bangladesh, Benin, Laos PDR, Cambodia, Democratic Republic of Congo (DRC), Myanmar, and Nepal.
RAVIN complemented existing efforts of the Ministry of Health and key partners in each focus country to facilitate the decision, planning, and introduction processes for the rotavirus vaccine. RAVIN’s diverse team of experts provided country-specific support to meet focus countries where they are in their decision-making process. RAVIN’s process started with stakeholder identification, proceeded to formal situational assessment to identify major barriers to rotavirus vaccine introduction, and culminated in the provision and mobilization of support to address identified barriers. The project goal was to increase attention to rotavirus vaccines in global and regional discussions and better leverage resources for sustainable country-based introduction.
IVAC led advocacy efforts to accelerate the decision-making process, and RAVIN built on existing relationships such as those with Gavi, WHO, and Unicef, to leverage existing resources. Other established partners included the Rotavirus Organization of Technical Allies (ROTA) Council, PATH, and the London School of Hygiene and Tropical Medicine (LSHTM) for costing and cost-effectiveness support. RAVIN served as a connector between key partners, linking systems and guiding the introduction and integration of rotavirus vaccines into routine immunization programs. RAVIN enhanced local capacity for the future and contributed to sustainable, equitable, and timely vaccine introduction and uptake for future generations.
JSI's technical assistance included developing quality introduction grant applications to Gavi, contributing to comprehensive introduction planning and roll-out of the vaccine in-country, and providing post-launch supportive supervision and monitoring. In addition, the project maximized opportunities at regional meetings and through its networks to support accelerated decision making and rotavirus introduction in other countries in the African and Asian regions.
As of 2022, five RAVIN countries have successfully introduced the vaccine into their routine childhood immunization programs.
Watch this video to learn more about how RAVIN combats preventable childhood deaths.
Watch this video to learn more about how communications propelled a successful rotavirus vaccine introduction in Benin.
Rotavirus Accelerated Vaccine Introduction Network (RAVIN) is a partnership between the Johns Hopkins Bloomberg School of Public Health International Vaccine Access Center (IVAC), JSI Research & Training Institute, Inc. (JSI), and the U.S. Centers for Disease Control and Prevention (CDC).